
In vitro human stem cell-based 3D testing systems
We generate reproducible iPS cell-derived 3D structures using rigorously optimized protocols. Each model is designed to meet the high standards of preclinical drug discovery, toxicity testing, and disease modeling.
Find the testing system that best suits your needs!
iPS cell-based three-dimensional spheroid systems
Human cerebellar organoid
3D human iPSC-derived model for studying cerebellar development, neurotoxicity, and disease mechanisms.
Human neural progenitor cell spheroid
Scalable in vitro model for neurodevelopmental and neurotoxicity testing.
Human cardiomyocyte spheroid
Human iPSC-derived cardiac model for assessing drug safety, cardiac function, and electrophysiological properties.
Human liver cell spheroid
3D hepatic model for investigating drug metabolism, enzyme activity, and assessing drug-induced liver injury (DILI) under physiologically relevant conditions.
Human mesenchymal stem cell spheroid
MSC-based 3D model for studying inflammation, fibrosis, tissue repair, and trilineage differentiation into bone, cartilage, and adipose tissue.
Human in vitro blood–brain barrier model
iPSC-derived co-culture system mimicking the human blood–brain barrier, enabling permeability, transport, and neurotoxicity studies.

How to Order and Prepare for a Test!
Preparing for Test Help Ensure All Goes Smoothly
To reduce your wait time, complete the personal information on the form in advance. Although most routine tests are covered under your provincial health insurance plan, some tests may not be covered with knowledge is power.
we connect directly with patients to deliver their results so they have valuable health information when they need it most, we care about our people.
-
Improving Through Automation
-
New Advanced Instruments
-
Strict Quality Practices
Find The Nearest Lab For You
Find the nearest location for you online through our contact page, or just call us.
Contact usMake An Online Appointment
Find the nearest location for you online through our contact page, or just call us.
Contact usCollect Sample And Send For Testing
Find the nearest location for you online through our contact page, or just call us.
Contact usVisit Our Patient Service Centre
Find the nearest location for you online through our contact page, or just call us.
Contact usGet Your Results Within 9 Hours
Find the nearest location for you online through our contact page, or just call us.
Contact usHelping To Deliver Answers For Health Questions!
Fixed Commitment To Excellence And Quality Of Our Test Results.
The insights we gain through testing then fuel our drug development practice, by identifying patterns and even individuals who might benefit from enrollment in specific drug trials.
With the support of our diagnostics capabilities to deliver drug trials that are both reliable.
Improving Your Health
Partnering for Through our investment in new high volume chemistry tech, we have brought next generation.
Better Health Outcomes
Building a healthier world clearly means looking at the future genetics and being ready for using it.
Innovations In Vaccines
Partnering for Through our investment in new high volume chemistry tech, we have brought next generation.
Trust Shared Purpose
Building a healthier world clearly means looking at the future genetics and being ready for using it.
Tips, News & Updates
Error: Contact form not found.
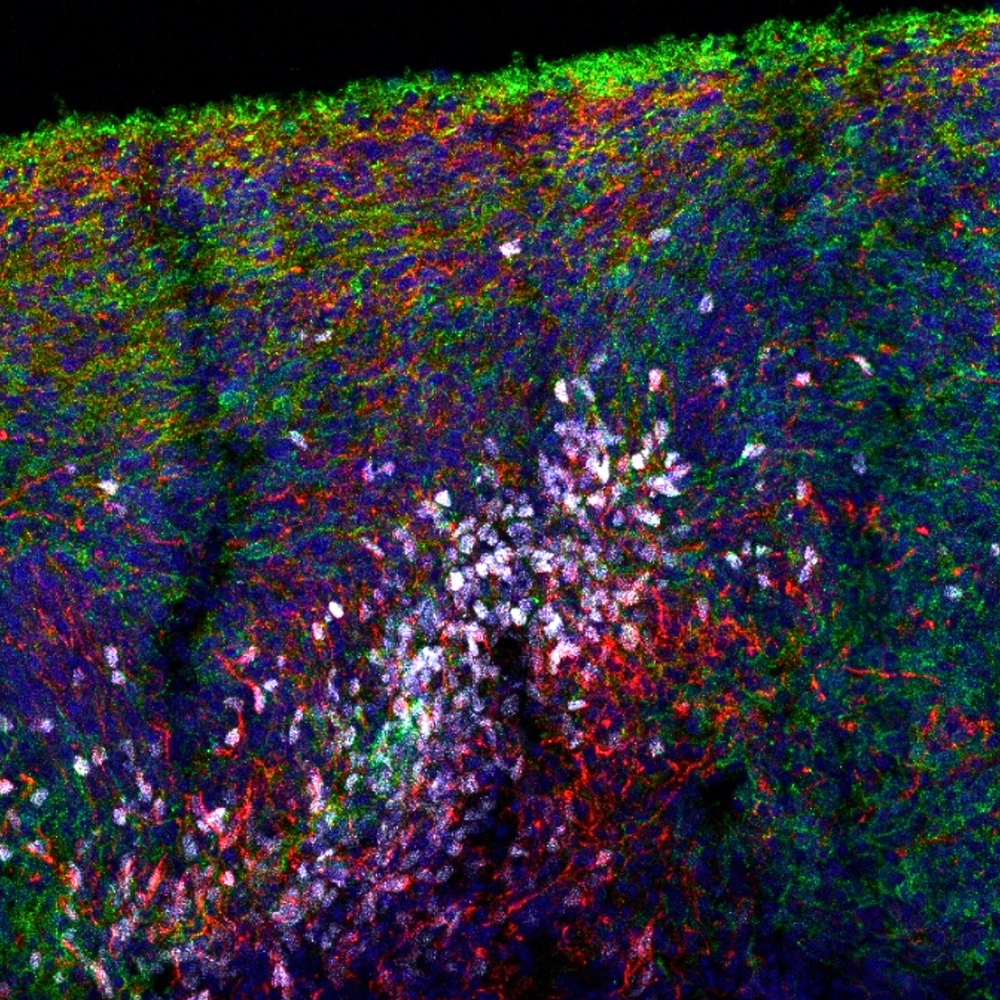
Human Cerebellar Organoids
Human induced pluripotent stem cell (iPSC)-derived cerebellar organoids offer a physiologically relevant 3D in vitro model for investigating motor coordination disorders, cerebellar development, neurodegenerative diseases, and neurotoxicology. These models capture essential aspects of cerebellar architecture and cell-type diversity, enabling detailed studies of neuronal differentiation, synapse formation, and disease-specific pathophysiology in a human context.
Applications include modeling genetic and acquired ataxias, congenital cerebellar malformations, assessing the effects of drugs or environmental toxins on cerebellar function, and exploring therapeutic strategies for restoring motor function. Their human-specific origin and reproducibility make them highly valuable for pharmaceutical screening, mechanistic research, and CNS-targeted therapeutic development.
Image: Early-stage cerebellar organoid immunostained for calbindin (green), Nestin (red), and SOX2 (white), with DAPI (blue) nuclear counterstain, illustrating an immature differentiation stage.
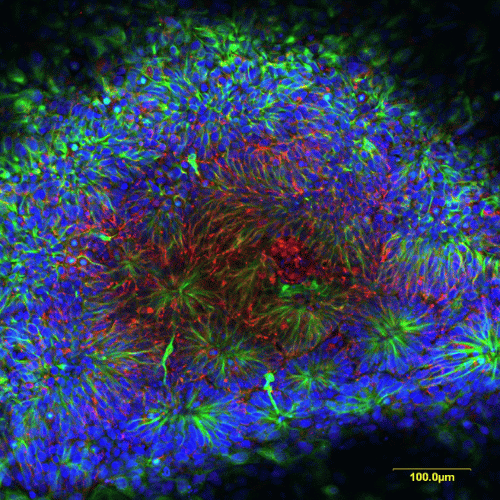
Human Neural Progenitor Cells
Human induced pluripotent stem cell (iPSC)-derived neural progenitor cell (NPC) spheroids provide a physiologically relevant in vitro platform for studying neurodevelopment, neurotoxicity, and disease mechanisms.
They enable assessment of neuronal differentiation, neurite outgrowth, and synaptogenesis - parameters not readily captured in conventional 2D cultures - with direct relevance for pharmaceutical, toxicological, and CNS-targeted drug research.
Typical readouts include high-content imaging, viability assays, calcium imaging, and gene expression profiling of lineage-specific markers. The model is available in 96- and 384-well formats, supporting medium-throughput applications and integration into automated workflows, while offering human-specific biological relevance.
Image: NPC cultures immunostained for Nestin (orange) and βIII-tubulin (green), with DAPI counterstaining for nuclei (blue).
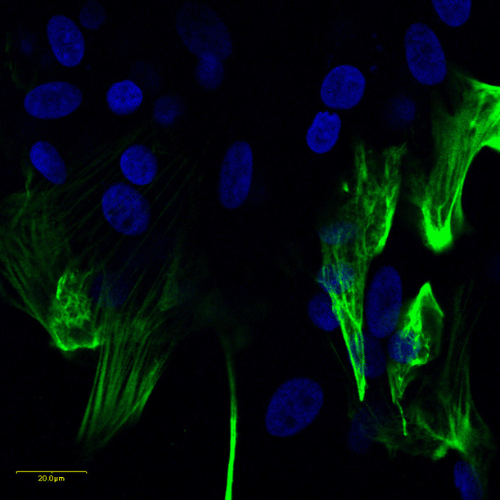
Human Cardiomyocytes
Human induced pluripotent stem cell (iPSC)-derived cardiomyocyte spheroids provide a physiologically relevant 3D model for assessing cardiac safety, drug efficacy, and disease mechanisms. They reproduce key structural and functional features of human myocardium, including synchronous contractility, ion channel activity, and electrophysiological responses, making them valuable for preclinical cardiotoxicity testing and for detecting pro- or antiarrhythmic drug effects.
Applications include safety pharmacology, functional cardiac profiling, and validation of cardiac-targeted therapies. Typical readouts involve microelectrode array (MEA) recordings, calcium transient imaging, beat rate variability analysis, and measurement of injury biomarkers such as troponin I. The model is compatible with 96-well formats, enabling medium-throughput testing with human-specific biological relevance.
Image: Pluripotent stem cells differentiated into functional cardiac cells. Cardiomyocytes were stained for cardiac troponin I (green), with nuclei counterstained with DAPI (blue).
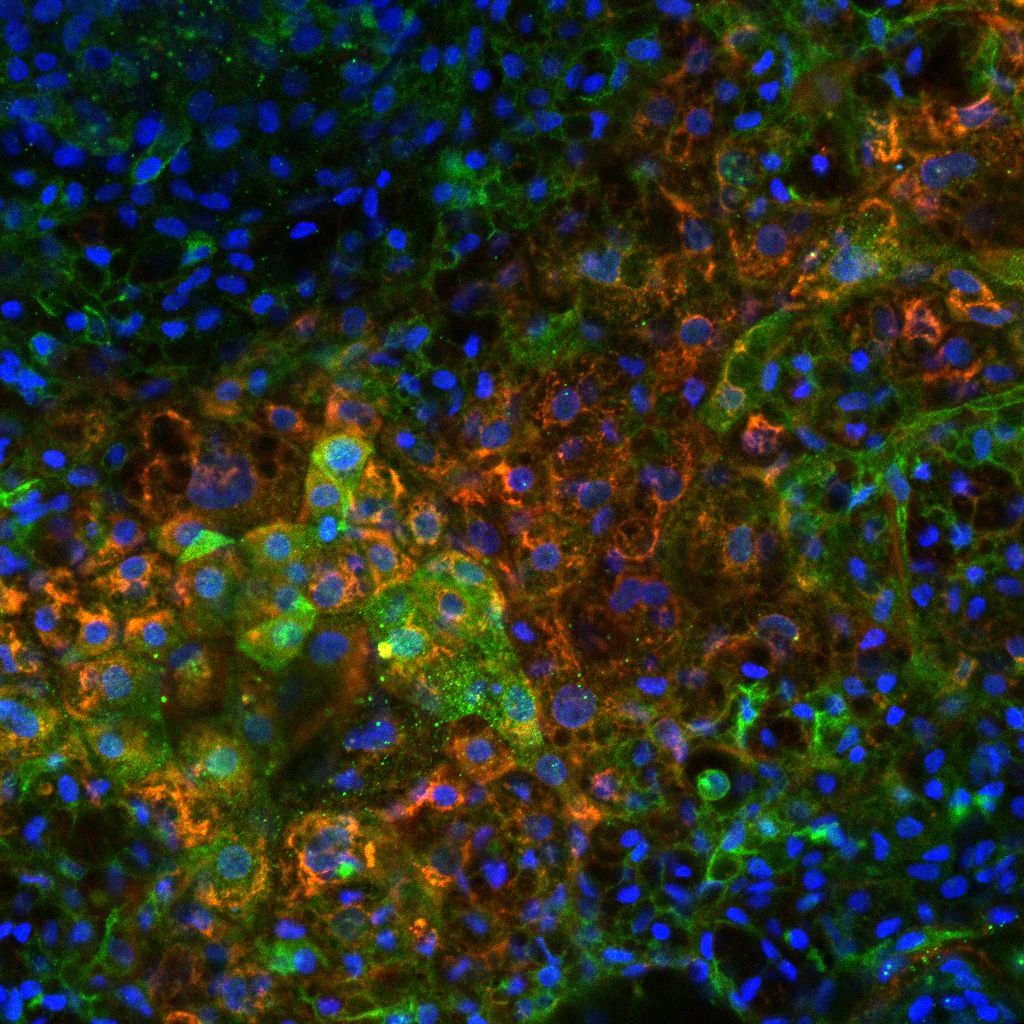
Human Liver Cells
Human liver cell spheroids derived from iPSC-hepatocytes offer a physiologically relevant 3D model for investigating drug metabolism, enzyme activity, and drug-induced liver injury (DILI). These models maintain key hepatic functions, including cytochrome P450 activity, bile acid metabolism, urea synthesis, and albumin secretion, providing predictive data for human liver responses that are often missed in 2D cultures or animal studies.
They are suited for safety assessment, efficacy testing, and mechanistic toxicology studies across pharmaceutical, chemical, and nutraceutical research. Typical readouts include enzyme activity assays, metabolic profiling, high-content imaging, and measurement of hepatotoxicity biomarkers such as ALT and AST. The model supports long-term culture in 96-well formats, enabling medium-throughput applications with strong translational relevance.
Image: Pluripotent stem cells expressing ABCG2 (green) differentiated into functional hepatocytes, immunostained for alpha-fetoprotein (orange), with nuclei labeled by DAPI (blue).
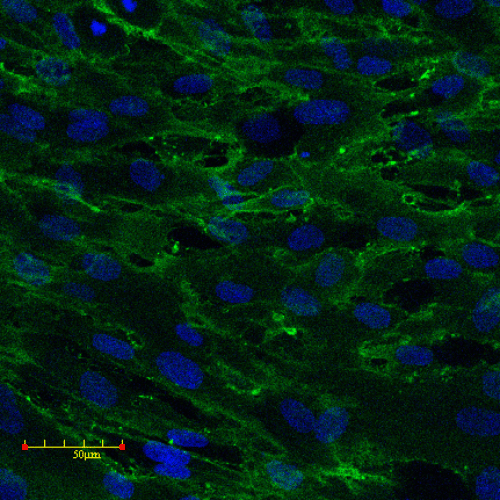
Human Mesenchymal Stem Cells
Human mesenchymal stem cell (MSC) spheroids derived from iPSCs provide a versatile 3D model for studying inflammation, fibrosis, tissue repair, and trilineage differentiation into bone, cartilage, and adipose tissue. Compared to primary MSCs, iPSC-derived MSCs offer superior reproducibility by eliminating donor-to-donor variability and are available in unlimited supply, ensuring consistent experimental performance across studies.
These models maintain the immunomodulatory and regenerative properties of MSCs within a controlled microenvironment, supporting applications in pharmaceutical development and regenerative medicine research. Typical readouts include cytokine profiling, extracellular matrix deposition analysis, high-content imaging, and lineage-specific gene or protein expression assays. The model is available in 96-well formats, enabling reproducible, medium-throughput testing with human-specific translational relevance.
Image: Pluripotent stem cells differentiated into mesenchymal stem cells (MSCs). MSCs were stained for CD44 (green), with nuclei counterstained with DAPI (blue).
Human Blood-Brain Barrier
The human in vitro blood–brain barrier (BBB) model is designed to recreate the tightly regulated interface between the circulatory system and the central nervous system. It is built from human iPSC-derived brain microvascular endothelial cells, astrocytes, and pericytes in a co-culture Transwell format, providing a consistent and reproducible alternative to primary cell-based systems. This eliminates donor-to-donor variability while enabling the assessment of barrier integrity, compound permeability, and transporter activity under physiologically relevant conditions.
Applications include early-stage CNS drug discovery, evaluation of neuroprotective strategies, and mechanistic studies of BBB disruption in neurological diseases. The system achieves high transendothelial electrical resistance (TEER) values within days of culture and supports long-term experiments for chronic exposure studies. Its isogenic composition ensures experimental consistency and facilitates integration with organ-on-a-chip platforms and other advanced in vitr

