Custom iPSC Generation from Human Donor Cells
Reprogramming somatic cells from both healthy donors and patients into high-quality, validated iPSC clones using footprint-free, integration-free methods — ideal for research, disease modeling, and drug discovery workflows.
3D Disease and Control Models from iPSCs
We generate 3D spheroid and organoid models from both healthy and patient-derived iPSCs. These models serve as powerful tools for academic and industrial research in disease modeling and therapy development, while helping to reduce reliance on animal testing.
CONTINUETesting Drug Safety & Efficacy in 3D Models
Human iPSC-derived 3D models provide physiologically relevant platforms for assessing compound safety and efficacy. Our assays support target validation, mechanism-of-action studies, and translational preclinical development.
CONTINUEGLP Laboratory Services
GLP-compliant testing services using human 2D and 3D cell-based models — designed for pharmaceutical, cosmetic, and medical device development. Our assays deliver reliable, high-quality data in line with regulatory standards.
Coming soon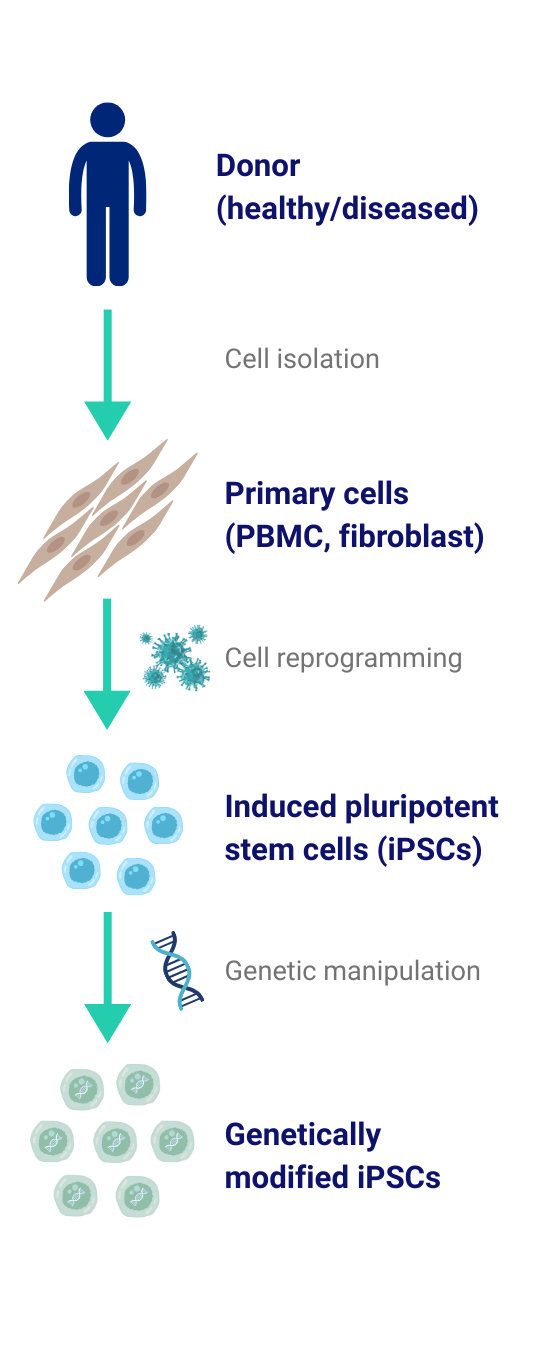
Custom iPSC Generation from Human Donor Cells
We generate custom, footprint-free induced pluripotent stem cell (iPSC) lines on request from any human donor - healthy or patient - and from the cell source you specify (e.g., PBMCs, dermal fibroblasts). Our integration-free reprogramming workflows are optimized for speed, reliability, and high clonality, delivering fully characterized lines ready for downstream use. If needed, we perform precise genome engineering with state-of-the-art editing systems - covering knock-outs, knock-ins, small edits, tag insertions, and targeted deletions - followed by clonal isolation and rigorous validation. Each line is supplied with a comprehensive QC package (identity and sterility checks, mycoplasma testing, karyotype/CNV assessment on request, pluripotency marker expression and tri-lineage potential), ensuring reproducibility and high quality for applications such as disease modeling, target validation, assay development, and drug discovery.
Related Research
Peer-reviewed publication demonstrating our expertise in pluripotent stem cell technologies.
Establishing a human embryonic stem cell clone with a heterozygous mutation in the DGCR8 gene
Reé D, Borsy A, Fóthi Á, Orbán TI, Várady G, Erdei Z, Sarkadi B, Réthelyi J, Varga N, Apáti Á.
Stem Cell Research (2020).
Read the full paper →
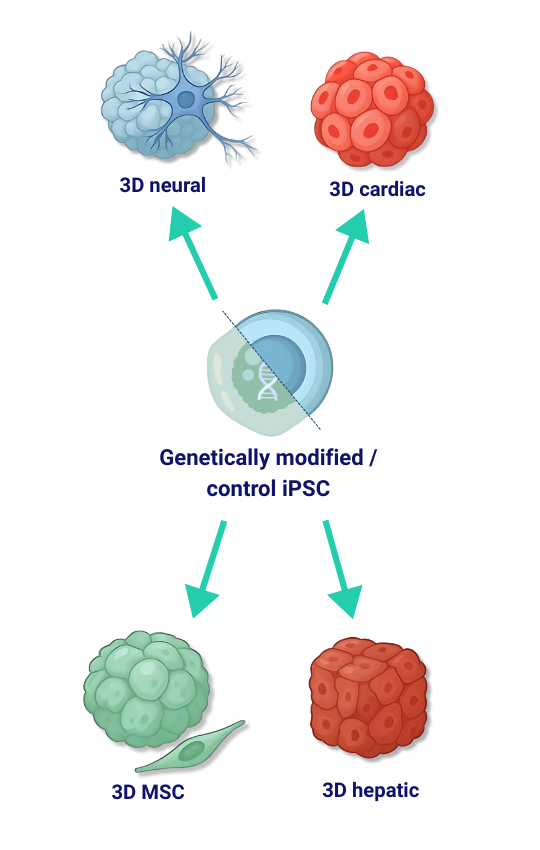
3D Disease and Control Models from iPSCs
We generate human iPSC-derived 3D spheroid and organoid systems – available as matched disease and isogenic control models – spanning neural, cardiac, hepatic and other tissue contexts. Unlike many providers, we maintain our own bank of proprietary, fully validated and legally compliant iPSC lines, available for commercial use, giving partners a unique, reliable source material for downstream applications. Built on standardized differentiation workflows, our platforms are optimized for reproducibility, scalability, and assay-readiness across screening and mechanism-of-action studies. On request, we engineer disease-relevant edits (knock-outs, knock-ins, point mutations, tag insertions, targeted deletions) and establish paired control lines, followed by clonal isolation and rigorous validation. Each 3D model is supplied with a comprehensive QC package (identity and sterility checks, mycoplasma testing, viability and uniformity metrics, morphology/size distribution, lineage-specific marker expression, and functional readouts where applicable), ensuring consistent performance for disease modeling, target validation, efficacy and safety assessment, and drug discovery – while reducing reliance on animal testing.
Related Research
Peer-reviewed publication demonstrating our expertise in iPSC-derived 3D models.
Functional Analysis of Antipsychotics in Human iPSC-Based Neural Progenitor 2D and 3D Schizophrenia Models
Farkas KG, Vincze K, Tordai C, Özgen Eİ, Gürler D, Deli V, Lilienberg J, Erdei Zs, Sarkadi B, Réthelyi JM, Apáti Á.
International Journal of Molecular Sciences (2025).
Read the full paper →
Testing Drug Safety & Efficacy in
3D Models
We perform advanced toxicology and mechanism-of-action studies on human iPSC-derived 3D neural, cardiac, and hepatic models — physiologically relevant systems that close the gap between in vitro assays and in vivo outcomes. Using high-throughput (HTS) and high-content (HCS) screening, we provide scalable and high-resolution assessments of compound safety, efficacy, and pharmacodynamics. Side-by-side comparisons with healthy and patient-derived models are supported by rigorous QC (identity, sterility, mycoplasma, morphology, viability, lineage-specific markers, and functional readouts), ensuring reproducibility and translational relevance while reducing reliance on animal models.
Related Research
Peer-reviewed publication demonstrating our expertise in iPSC-based safety and efficacy testing.
Generation of multidrug resistant human tissues by overexpression of the ABCG2 multidrug transporter in embryonic stem cells
Erdei Z, Schamberger A, Török G, Szebényi K, Várady G, Orbán TI, Homolya L, Sarkadi B, Apáti Á.
PLoS One. 2018 Apr 12;13(4):e0194925. doi: 10.1371/journal.pone.0194925. PMCID: PMC5896897
Read the full paper →
Contact us for more information!
Innocell Kft.
1119 Budapest, Than Károly utca 20.
E-mail: info@innocellclinic.com
