Building the Future of Regenerative Medicine
"Looking back, the creation of InnoCell Clinic is closely linked to a clinical trial conducted many years ago at the Kelen Hospital in Budapest. At our hospital, we treated patients, most of whom suffered from severe diabetes. Many of them were facing limb amputation and were classified as “No Option” cases. They lived their days in crippling pain, and many were forced to stop after just a few steps.
Through stem cell therapy — which was clearly innovative at the time — we witnessed something extraordinary with our own eyes: a new collateral vascular network formed around the blocked vessels, blood circulation restarted, pain was significantly reduced, and in most cases amputation became avoidable. This experience, which was almost unbelievable at the time, revealed the extraordinary potential of stem cells.
The subsequent discovery of induced pluripotent stem cells (iPSCs) opened up even broader possibilities for regenerative medicine. Together with Prof. Balázs Sarkadi, one of the most respected figures in stem cell research worldwide, we launched InnoCell Clinic with the vision of applying these miraculous cells to real medical interventions.
Today, InnoCell Clinic focuses on the development of advanced in vitro platforms — human stem cell preparations, 3D spheroids, and disease models — that support pharmaceutical, medical device, and cosmetic research. These models provide reliable, human-specific insights, saving both time and resources, while eliminating the need for animal testing. This transition is increasingly encouraged and supported by regulatory authorities such as the FDA and EMA.
Our mission is to create reliable, proven technologies for preclinical testing and drug development, led by internationally recognized experts with decades of experience in healthcare, pharmaceuticals, and stem cell research. At InnoCell Clinic, we believe that research in this field must go hand in hand with strong ethical principles, offering next-generation tools that not only drive innovation forward, but also carry a deep sense of responsibility towards patients."
Zsuzsanna Kövesd, MD
Managing Director

Advanced Human Cell-Based Models
InnoCell was established to play a leading role in the dynamic growth of the biotechnology industry. Our laboratory and services stand out, built on globally benchmarked, high-level technology that supports next-generation biomedical research and testing.
Our core focus lies in the production of human stem cell preparations, stem cell-based 3D constructs (spheroids/organoids), and disease models. These platforms serve the pharmaceutical and cosmetic industries, providing advanced, human-relevant tools for research and development. In health-related fields in particular, such approaches are driving meaningful progress. The use of human cells in preclinical efficacy and toxicology testing is increasingly replacing animal models.
At InnoCell, we prioritize scientific progress that is both ethical and sustainable. We support the transition to human-relevant testing methods that align with evolving international guidelines. Regulatory bodies like the FDA, EMA, and China’s NMPA increasingly advocate the use of human cell-based systems in preclinical drug development and toxicology to reduce animal testing [EMA/CHMP/CVMP/JEG-3Rs/450091/2012, 15 December 2016]. In the cosmetics industry, such testing is already banned.
We believe innovation should reflect both scientific and societal responsibility. Our mission is to develop high-quality, human cell-based platforms that support this shift and contribute to a more forward-looking, ethical, and predictive approach to research and development.
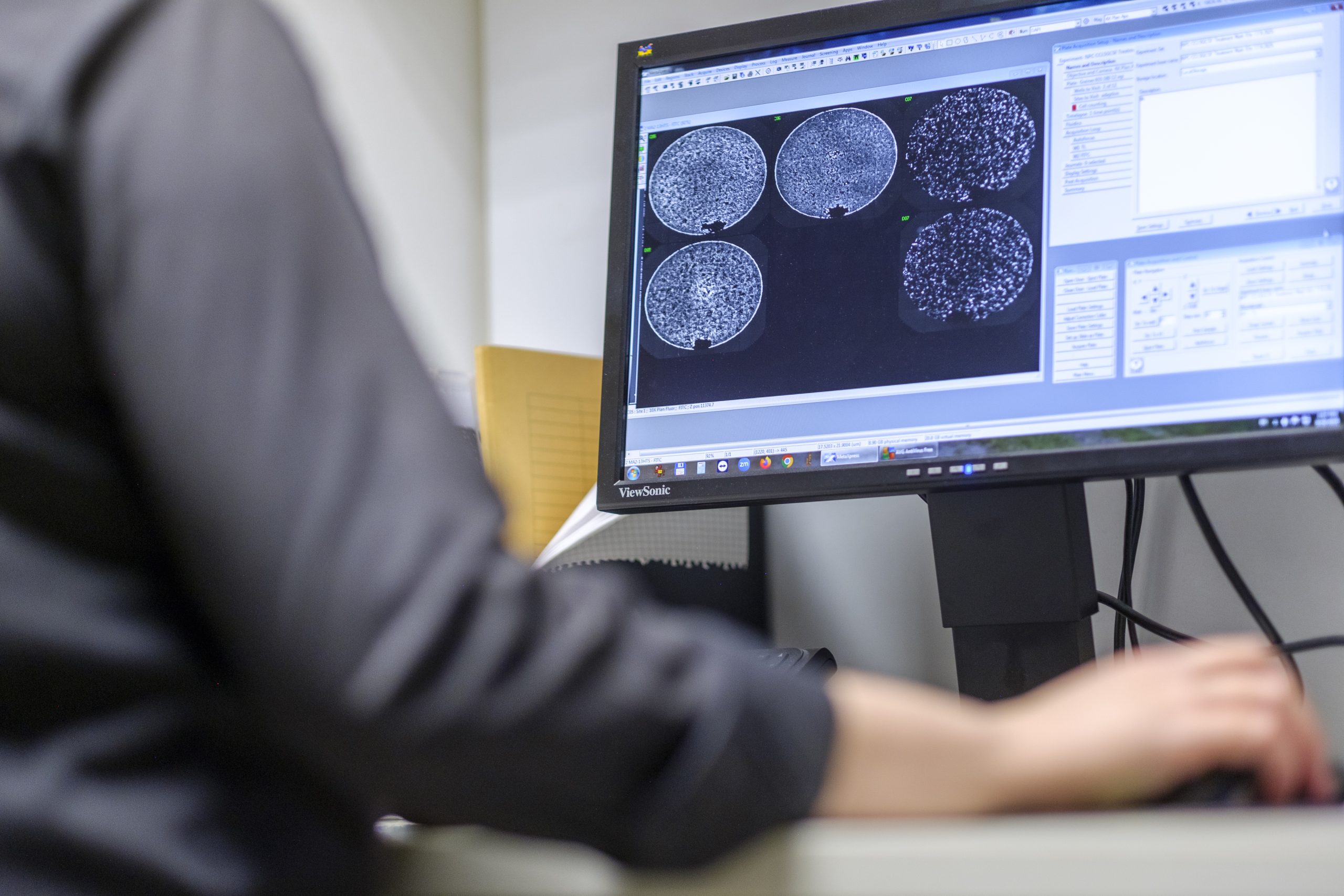
Unique Technology and Expertise
Pluripotent stem cells have the unique ability to differentiate into virtually any human tissue, making them a powerful foundation for building advanced preclinical models. From neural, cardiac, and hepatic systems to complex disease models, stem cell–derived 3D cultures are increasingly replacing traditional animal models in efficacy and toxicology studies.
The development of appropriate culturing, differentiation, and genetic modification technologies is currently underway. However, the stable, certified, and appropriately licensed production of human stem cell preparations is already a fundamental requirement for regulatory acceptance in preclinical studies.
At InnoCell, we have undertaken the task of establishing these highly reliable technologies for preclinical testing and drug development. Our work is backed by a team of internationally recognized experts with decades of experience in healthcare, the pharmaceutical industry, and stem cell research — ensuring the depth, precision, and reliability needed to meet evolving scientific and regulatory standards.

Our Partners with major grant supports
Salus Ltd., Budapest, Hungary
Manufacturer and distributor of medical devices and orthopedic equipment, with a portfolio covering rehabilitation and home care.
HUN-REN Biological Research Centre, Szeged
National life sciences institute with over 260 scientists, active in molecular and cell biology, human health, and environmental research.
Our Partners with research contracts
Neurovascular Unit & Therapeutic Innovation Laboratory, CEA, Paris
Part of the French Alternative Energies and Atomic Energy Commission (CEA), with collaboration focused on brain organoid development.
Anthelos Ltd.
Provides research tools for drug discovery and molecular diagnostics, with focus on early detection and treatment monitoring.
Bioxol Ltd.
Develops generic reagents and molecular biology solutions, with additional commitment to training young researchers.


Providing High Quality Test Services For Your Health!
Helping To Deliver Answers For Health Questions.

Appointments may be booked to help reduce your wait time. If you are unable to book an appointment, we do our best to minimize your wait, but the length of your visit will depend on the number of patients in the Patient Service Centre. Typically, wait times are a little longer in the morning as patients tend to fast for tests over night.
To provide a comfortable and safe environment for our patients and employees, please avoid wearing scented perfumes or creams when visiting our Patient Service Centres.
World Class Diagnostics
We have developed some of the world's most advanced testing capabilities to help improve health and lives.
Leader in Drug Development
With the support of our diagnostics capabilities to deliver drug trials that are both reliable and rigorous.
We process more than 3 million patient specimens per week and help medical professionals and patients make important health decisions, by identifying patterns and even individuals who might benefit from enrollment in specific drug trials.



Tips, News & Updates
Error: Contact form not found.
